Some Basic Principles and Techniques — Revision Notes
NEET-UG · Chemistry
Quick revision notes for Some Basic Principles and Techniques — key concepts, formulas, and definitions for NEET-UG Chemistry preparation.
Interactive on Super Tutor
Studying Some Basic Principles and Techniques? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for NEET-UG Chemistry.
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedRevision Notes — Some Basic Principles and Techniques
Key concepts, formulas, and definitions from Some Basic Principles and Techniques for NEET-UG Chemistry preparation.
Key Topics to Revise
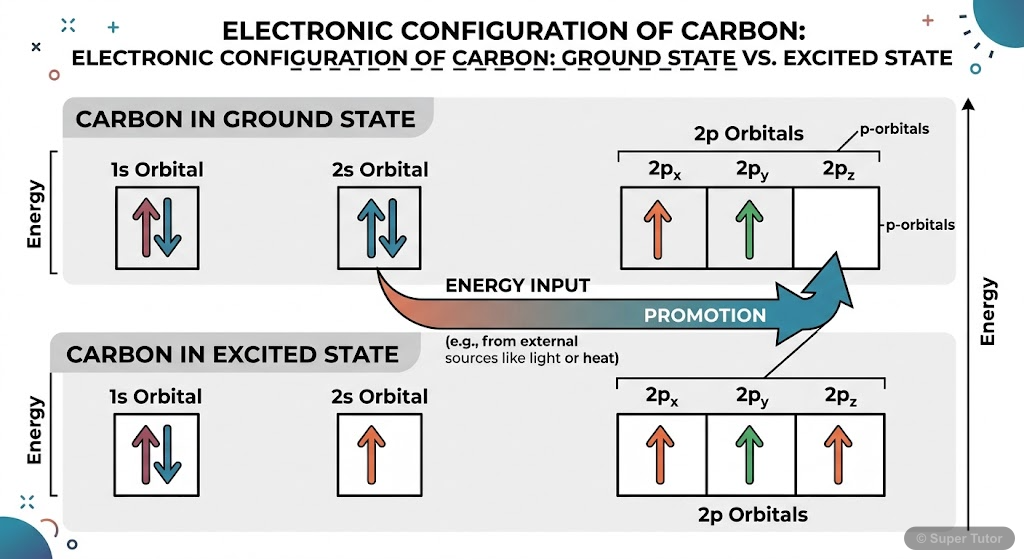
Tetravalency and Hybridization of Carbon
- Carbon has 4 electrons in outer shell (1s² 2s² 2p²), making it tetravalent
- Ground state carbon is divalent, but promotion of 2s electron to 2p orbital makes it tetravalent
- Promotion requires 96 kcal/mol energy, which is regained during bond formation
IUPAC Nomenclature System
- IUPAC name = Prefix + Word root + Primary suffix + Secondary suffix
- Word root indicates number of carbon atoms (meth-, eth-, prop-, but-, etc.)
- Primary suffix indicates saturation: -ane (single bonds), -ene (double bonds), -yne (triple bonds)
Structural Isomerism
- Same molecular formula but different structural arrangements
- Five types: Chain, Position, Functional, Tautomerism, Metamerism
- Chain isomerism: Different carbon skeletons (n-butane vs isobutane)
Stereoisomerism
- Same molecular and structural formula but different spatial arrangement
- Three types: Conformational, Optical, Geometrical
- Conformational: Due to rotation around single bonds (staggered vs eclipsed)
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
Frequently Asked Questions
What topics are covered in Some Basic Principles and Techniques for NEET-UG?
How important is Some Basic Principles and Techniques for NEET-UG?
How to prepare Some Basic Principles and Techniques for NEET-UG?
More resources for Some Basic Principles and Techniques
Syllabus
Some Basic Principles and Techniques — syllabus
Important Topics
Some Basic Principles and Techniques — important topics
Practice Questions
Some Basic Principles and Techniques — practice questions
Study Plan
Some Basic Principles and Techniques — study plan
Formula Sheet
Some Basic Principles and Techniques — formula sheet
For NEET-UG aspirants
Get the full Some Basic Principles and Techniques chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for NEET-UG Chemistry.