Atoms
Kerala Board · Class 12 · Physics
Summary of Atoms for Kerala Board Class 12 Physics. Key concepts, important points, and chapter overview.
Interactive on Super Tutor
Studying Atoms? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for chapter summary and more.
1,000+ Class 12 students started this chapter today
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedOverview
The chapter 'Atoms' explores the fundamental structure of matter at the atomic level, tracing the historical development from Thomson's plum pudding model to Bohr's quantum mechanical model. This chapter is crucial for understanding how atoms are structured, why they emit characteristic spectra, and
Key Concepts
Proposed in 1898
Proposed in 1898, this model suggested that atoms consist of a uniformly distributed positive charge with electrons embedded like seeds in a watermelo
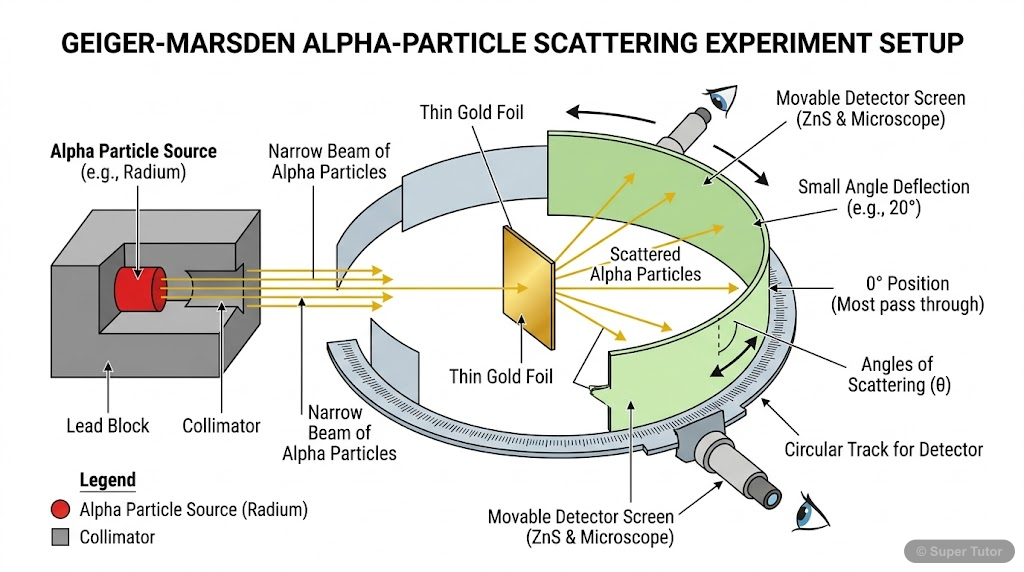
Conducted by Geiger and Marsden
Conducted by Geiger and Marsden in 1911, this experiment involved bombarding a thin gold foil with alpha particles. Most particles passed through, but
This model proposed that the entire
This model proposed that the entire positive charge and most of the atom's mass are concentrated in a tiny nucleus (10^-15 to 10^-14 m), with electron
According to classical electromagnetic theory
According to classical electromagnetic theory, revolving electrons should continuously emit radiation, lose energy, and spiral into the nucleus, makin
First postulate
First postulate: Electrons revolve in certain stable orbits without emitting radiation (stationary states). Second postulate: Angular momentum is quan
Learning Objectives
- Understand the historical development of atomic models from Thomson to Rutherford to Bohr
- Explain Rutherford's alpha-particle scattering experiment and its significance in discovering the nucleus
- Comprehend the limitations of classical physics in explaining atomic stability and spectra
- Master Bohr's three postulates and their application to hydrogen atom
- Calculate energy levels, orbital radii, and transition frequencies in hydrogen atom
Frequently Asked Questions
What are the important topics in Atoms for Kerala Board Class 12 Physics?
How to score full marks in Atoms — Kerala Board Class 12 Physics?
Sources & Official References
- Kerala Board of Public Examinations — keralapareekshabhavan.in
- National Education Policy 2020 — education.gov.in
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Atoms
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Atoms chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Kerala Board Class 12 Physics.